Thermodynamic Relationships - Problem Set 8 Solution Key - Fall 2014 | CHEM 3615 | Exams Physical Chemistry | Docsity

What is the major factor that accounts for most of the difference in these two values of pressure (ideal gas law vs. van der Waals equation)? | Socratic

The van der Waal's equation of state for some gases can be expressed as : (P + (a)/( V^(2))) ( V - b) = RT Where P is the pressure , V








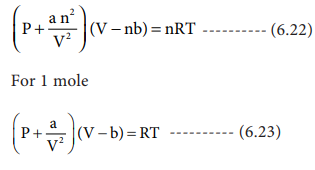
![PDF] A Modified Form of the van der Waals Equation of State PDF] A Modified Form of the van der Waals Equation of State](https://i1.rgstatic.net/publication/253282197_A_Modified_Form_of_the_van_der_Waals_Equation_of_State/links/545ba2d90cf2f1dbcbcafdda/largepreview.png)


![Solved P= aN2 V2' 2-2. [20 points] Using the van der Waals | Chegg.com Solved P= aN2 V2' 2-2. [20 points] Using the van der Waals | Chegg.com](https://media.cheggcdn.com/media/eeb/eebc5207-da0f-4603-8b84-5abbdf4904c6/phpBa9k8H.png)

